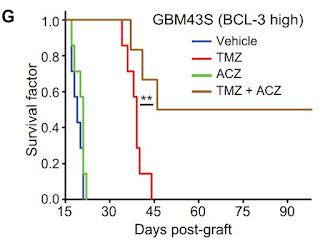
FRom supplementary info of this Article
https://www.sciencedirect.com/science/article/pii/S0304383518303124?viewFullText=true#mmc1
Presentation of the main achievements in preclinical studies for GBM,
contemplating detailed information such as IC50, IC90,
EC50 and LD50 values for repurposed drugs.
|
Drug
|
Main achievements
|
New IC50
|
References
|
Chloroquine
|
Chloroquine treatments (10
and 25 μΜ) halved proliferation of primary cultures from GBM specimens and
cell lines (U 373 and U 87). Chloroquine inhibited MMP-2 activity and
GBM invasion; In an MTT assay, U251, LN229, and U87 glioma cell lines were
treated with increasing concentrations of chloroquine for 48 hours (10‑100 μM).
|
30 μΜ (U251, LN229, U87)
40 μΜ (U251‑TMZR,
LN229‑ TMZR, U87‑ TMZR)
|
[1,2]
|
Hydroxichloroquine
|
Hydroxichloroquine killed
glioma cells that were highly resistant to temozolomide, proving its
cytotoxicity. Quinoline-based antimalarial compounds are cytotoxic to glioma
cells.
|
-
|
[2]
|
Mefloquine
|
Mefloquine effectively
killed U251 cells at much lower concentrations than chloroquine. Mefloquine
killed U 251 and U 251-TMZ resistant cells in a concentration dependent
manner.
|
10 μΜ (U251, LN229, U87)
15 μΜ (U251‑ TMZR,
LN229‑ TMZR, U87‑ TMZR)
|
[2]
|
Quinacrine
|
Quinoline-based antimalarial
compounds are cytotoxic to glioma cells.
In an MTT assay, the U251 and U251-TMZR
glioma cell lines were treated with increasing concentrations of quinacrine
(10‑100 μΜ) for 48 hours. In a
subcutaneous human xenograft U87 glioma model, nude mice were treated with 50
mg/kg of quinacrine and tumors were harvested after 24 hours. Quinacrine
significantly reduced tumor progression.
|
5 μΜ (U251, LN229, U87)
8 μΜ (U251‑TMZR, LN229‑TMZR,
U87‑TMZR)
|
[2,3]
|
Pyrvinium pamoate
|
GBM samples with a CD133 high
fraction are much more sensitive to pyrvinium treatment than those with a
CD133 low fraction. CD133+ cells decline upon treatment with 200 nmol/L with
pyrvinium for 48 hours in both primary (BT428) and recurrent (BT 566) GBM
samples. Treated cells with pyrvinium at its IC80 levels for
3 days were intracranially injected in immunodeficient mice, that
later displayed no evidence of tumor formation.
|
239.8 nmol/L (BT241), 122.5 nmol/L (BT486)
|
[4]
|
Mebendazole
|
Oral administration of
mebendazole at 50 mg/kg from day 5 after tumor implantation in C57BL/6 mice
slowed tumor growth. 100 mg/Kg daily led to toxicities. 60919 GBM cells were
incubated with 0.1 or 1 μM of mebendazole, and 1μM of mebendazole clearly
reduced the polymerization of tubulin; this activity of mebendazole was also
verified at 0.1 or 0.2 μM after 72 h
|
0.24 μM (GL261),
1μM (060919 GBM)
|
[5]
|
Acyclovir
|
Acyclovir at high
concentrations (up to 500 mg/mL) inhibited growth in tissue culture of the
human glioblastoma cell lines T98G, SNB-19, and U 373 by as much as 68.3%.
|
-
|
[6]
|
Ritonavir
|
In vitro, ritonavir induced a
G1-block at the 100-μM dose in GL15 cells. Rats were treated daily with 40
mg/kg, IP, until their death but there was no control over tumor growth, most
likely because the therapeutic dose was not reached in the tumor.
|
-
|
[7,8]
|
Atazanavir
|
50 μmol/L atazanavir induced
inhibitory effects in both U 251 and LN229 cells.
|
-
|
[9]
|
Ribavirin
|
Ribavirin treatment (30 μM)
leads to a significant decrease in all glioma cell growth. Ribavirin
treatment in vivo significantly
enhances chemo-radiotherapy efficacy and improves survival of rats and mice
orthotopically implanted with gliosarcoma tumors or glioma stem‑like cells,
respectively.
|
53.6 μM (A 172)
27.9 μM (AM-38)
55.0 μM (T98G)
59.7 μM (U87)
664.2 μM (U 138)
257.7 μM (U 251)
76.9 μM (YH-13)
|
[10]
|
Itraconazole
|
2-80 μM of itraconazole led
to cytotoxicity in U87 and C6 cells. Nude mice with U87 subcutaneous tumor
xenografts were treated with 75 mg/kg, twice daily, by gavage for 3 weeks.
Itraconazole inhibited the proliferation of glioblastoma cells both in vitro and in vivo.
|
-
|
[11]
|
Ciprofloxacin
|
Ciprofloxacin induced tumor
cell death in a dose-dependent manner. IC50 reduced to 22.8 μM for
ciprofloxacin in the presence of 62.5 μM of temozolomide.
|
259.3 μM (A 172)
|
[12,13]
|
Salinomycin
|
Salinomycin significantly
reduced the cell viability of GL261 neurospheres and GL261 adherent
cells in a dose-dependent manner. The inhibitory effect was more effective
than that of 1-(4-amino-2-methyl-5-pyrimid
l)-methyl-3-(2-chloroethyl)-3-nitrosourea hydrochloride and vincristine.
Salinomycin depleted GL261 neurospheres from tumorspheres and induced
cell apoptosis. In addition, it prolonged the median survival time of
glioma-bearing mice.
|
-
|
[14]
|
Minocycline
|
50 μM of minocycline reduced
cell viability of U 87, U 251 and C6 glioma cells. Alone, it did not affect cell
viability of normal cells (SVGP12 and rat primary astrocytes).
Mice that were injected with
C6 cells and treated with minocycline (20 or 100 mg/kg, IP) in saline, daily,
during 10 days had a slower tumor growth rate when compared to control group.
|
30 μM (C6)
|
[15]
|
Doxycycline
|
Doxycycline exerted mild
anti-proliferative effects after high‑concentration treatments (20, 25, 30,
and 35 µg/ml) on glioma cell lines U 251HF, U 87 and LN229.
|
-
|
[16]
|
Tigecycline
|
Tigecycline inhibited glioma
cell growth in a concentration-dependent way. 10 μM of tigecycline alone did
not affect cell viability of normal cells (SVGp12 and rat primary
astrocytes).
Tigecycline effectively
inhibited tumor growth in the xenograft tumor model of U87 glioma cell, after
the administration of tigecycline (100 mg/kg in DMSO), daily, for 10 days.
|
-
|
[17]
|
Chlorpromazine
|
A significant decrease in
cell viability was observed in cells treated for 24h with high concentration
of chlorpromazine (≥20 μM). Overall survival significantly improved for U
251-TMZR orthotopic mouse brain tumor models, but not for the U
251 group, after treatment with 5 or 7 mg/Kg, IP for three times a week for 2
weeks and 5 days after tumor implantation.
|
18.8-27.7 μM (C6)
15 μM (SH-SY5Y)
|
[18,19]
|
Thioridazine
|
GBM8401 cells were treated
with at concentrations ranging from 5 to 15 μM for 24h. GBM8401 cells
were treated concentrations of 10 and 20 μM for 24 h. Thioridazine
induces autophagy in GBM8401 and U 87 cells, and has cytotoxic effect at 7.5 μM.
U 87 cells were subcutaneously implanted into mice. Thioridazine
(5 mg/kg/day, 5 days/week, IP) significantly reduced tumor size. Flow
cytometry of propidium iodide-stained glioma cells treated with thioridazine,
fluphenazine, or perphenazine (6–50 µM) resulted in a concentration-dependent
increase of fragmented DNA up to 94% vs 3% in controls by all agents, with
thioridazine being the most potent.
|
13.7 μM (C6)
11 μM (SH-SY5Y)
|
[20,21]
|
Fluphenazine
|
Fluphenazine, from 0-24 μM, induced a marked and concentration-dependent decrease in cell
viability in glioma C6 cells. Flow cytometry of propidium iodide-stained
glioma cells treated with thioridazine, fluphenazine, or perphenazine (6–50
µM) resulted in a concentration-dependent increase of fragmented DNA up to
94% vs 3% in controls by all agents.
|
19-24.5 μM (C6)
15 μM (SH-SY5Y)
|
[21]
|
Perphenazine
|
Perphenazine, from 0-24 μM, induced a marked and concentration-dependent decrease in cell
viability in glioma C6 cells. Flow cytometry of propidium iodide-stained
glioma cells treated with thioridazine, fluphenazine, or perphenazine (6–50
µM) resulted in a concentration-dependent increase of fragmented DNA up to
94% vs 3% in controls by all agents.
|
15.8 μM (C6)
15 μM (SH-SY5Y)
|
[21–23]
|
Olanzapine
|
Treatment
with olanzapine (up to 100 μM) inhibits the
proliferation of established glioblastoma cell lines and enhances the
antiproliferative effect of temozolomide on U 87 and A 172 cells.
|
27.9 μM (A172)
49.1 μM (U87)
|
[24]
|
Penfluridol
|
With
increasing concentrations, penfluridol significantly suppressed the growth of
several glioblastoma cell lines in a concentration and time-dependent manner.
Penfluridol (10 mg/kg by oral gavage, daily) led to a 65% suppression of glioblastoma
tumor growth (U 87) in mice.
|
2–5 μM (GBM43, GBM10, GBM44,
GBM28, GBM14, T98G, U 251, U 87, SJ‑GBM2, CHLA-200)
|
[25]
|
Quetiapine
|
Relatively
high doses of quetiapine (>25 μM) may inhibit cell proliferation by
retarding cell cycle in the G2-M phase. In xenograft tumor model in nude
mice, quetiapine (20mg/kg, IP) alone or combined with temozolomide,
significantly suppressed tumor growth, displaying a synergistic antitumor
effect with TMZ.
|
-
|
[26]
|
Lithium
|
Inhibition
of migration was dose-dependent, with a near complete blockade at 20 mM
lithium for X12 (human biopsy) and U 87 glioma cells. A reduction in
viability of about 20% was seen after 48 h of 20 mM lithium treatment in
U 87 cells. Lithium concentrations above 5 mM can affect the
proliferation, apoptosis and migration of glioma cells via GSK-3 inhibition.
Combination with 1.2 mM Lithium potentiated TMZ-induced cell death in TP53wt
glioma cells. TMZ combined with Lithium prevented tumor growth in vivo and increased mice median
survival times.
|
-
|
[27,28]
|
Donepezil
|
Treatment of Hs683 cells
with 2.5 μM donepezil for 72 h blocked a large majority of Hs683 cells in
division, as also observed for T98G and U373 cells. Mice with orthotopically
implanted Hs683 cells and treated with donepezil + TMZ (2 mg/kg + 40
mg/kg, per os, thrice a week,
respectively) had a significant increase in survival, while treatment with
donepezil alone (2 mg/kg per os,
thrice a week) did not show significant benefits.
|
-
|
[29,30]
|
Memantine
|
Memantine (up to 600 μM) had
an antiproliferative effect on T98G cells, but not on U 251 cells.
|
400 μM (T98G)
|
[31]
|
Paroxetine
|
Paroxetine induced a
dose-dependent decrease in cell viability. Concentrations of SSRIs that
induced apoptosis are higher than those achieved with the current therapeutic
use of these drugs
|
12–30 μM (C6)
|
[32]
|
Fluoxetine
|
25-50 μM fluoxetine application decreased the viability of various glioma cell
lines. The concentrations of SSRIs that induced apoptosis are higher than
those achieved with the current therapeutic use of these drugs.
|
12–30 μM (C6)
|
[23,32,33]
|
Sertraline
|
Sertraline alone (up to 10 μM) displayed cytotoxicity in U 87 cells. When
combined with imatinib or temozolomide, the antiproliferative effect was
markedly improved.
|
3.1-6.8 μM (U 87)
|
[23]
|
Fluvoxamine
|
The inhibitory effect of
fluvoxamine on actin polymerization was concentration dependent. 20-30 μM was enough to inhibit lamellipodia formation and migration and invasion
of U 87 and U 251 cells in
vitro. Therapeutic doses of fluvoxamine were sufficient to prevent
invasion of GBM cells (A 172,
U 87, and U 251). Daily administration of fluvoxamine (50 mg/kg) inhibited GBM cell
invasion and prolonged survival in mice bearing GBM tumors.
|
30 μM
|
[34]
|
Imipramine
|
Exposure to imipramine
60 μM for 7 days strongly
reduced the ability of U 87 and C6 cells, but not primary cultured rat
astrocytes, to form colonies, due to cell death; 10 μM imipramine inhibited
mitochondrial activity at a rate dependent on the oxygen content in the
atmosphere (from 6% in hypoxia, 11% in average hypoxia, and 19% in hypoxia‑reoxygenation
to 26% in 20% oxygen).
|
-
|
[35–37]
|
Amitriptyline
|
10 μM amitriptyline
inhibited mitochondrial activity on TG98 cells at a rate dependent on the
oxygen content in the atmosphere (from 6% in hypoxia, 11% in average hypoxia,
and 19% in hypoxia-reoxygenation to 39% in 20% oxygen). Low-dose amitriptyline (0.14-0.5 mM) has emerged as a potential strategy for inducing inhibition
of cellular respiration in tumor cells.
|
-
|
[38,39]
|
Escitalopram
|
Significant
decreases in the proliferation of C6 glioma cells were detected with the
increase in the escitalopram concentration and incubation period. Comparing
to controls, cell proliferation after 24 h of incubation were 97.7,
85.9, 74.5 and 67.9% for 25, 50, 100 and 200 μM escitalopram,
respectively. After 48 h, it was found to be 96.5, 68.0, 50.7 and 39.9% for
25, 50, 100 and 200 μM concentrations, respectively. Results indicate escitalopram induced
citotoxitcity and apoptotic events in C6 glioma cells.
|
106.97 μM (C6)
|
[40]
|
Levetiracetam
|
40, 80, 160, and
360 μg/mL reduced T98G cell numbers to a certain degree relatively to the number of control cells (at 72h);
80, 160, and 360 μg/mL reduced A 172 cell numbers by 20.2%, 23.5%, and 24.8% relatively to the number of control cells.
|
-
|
[41]
|
Valproic acid
|
5 to 20 mM induced G2/M cell
cycle arrest and increased the production of ROS (in U 87, GBM8401, and
DBTRG-05MG GBM‑derived cell lines); it inhibited MTT dehydrogenase activity
at concentrations over 800 µM (in those 3 GBM cell lines); Epilepsy patients
generally accumulate total plasma concentrations in the range of 0.3-0.7 mM,
when treated orally with 15-20 mg/kg valproic acid per day (lower concentrations of around 40-200 µM at a daily
treatment are observed in the brain indicating valproic acid brain/plasma
ratios in the range of 0.07 to 0.28); a dose escalation study in GBM patients
defined 60 mg/kg valproic acid per
day as maximal tolerable dose which gave rise to median plasma concentrations
of about 0.85 mM and a maximal plasma concentration not exceeding 1.4 mM.
|
1.4 mM (T98G)
1.0 mM (U 251)
1.3 mM (U 87)
|
[42,43]
|
Propofol
|
5 and 10 μg/ml
significantly inhibited the proliferation of U 373 glioma cells at 48 and 72
h.
|
-
|
[44]
|
Disulfiram
|
Classically TMZ
resistant cells (SF188 cells) were sensitive to 500 nM, a sufficient
concentration to suppress growth in monolayer by approx. 100% over 72 hours;
U 251 cells treated with 200 nM were suppressed in growth by 80% and 500 nM
doses completely eliminated the cells; IC90 value reported for
disulfiram in SF188 cells was 100 nM.
|
-
|
[45,46][
|
Dimethyl fumarate
|
It is rapidly metabolized
to MMF and has a Cmax in plasma of ∼15 μM, with an
approximate steady state tissue and plasma concentration of 5 μM. GBM cells
were treated with MMF (5 μM), enhancing toxicity of velcade and carfilzomib.
|
-
|
[47]
|
Digitoxin
|
Non-cytotoxicity
concentration (20 nmol/l) can induce TRAIL‑mediated apoptosis of GSCs; While
digitoxin was capable of inhibiting HIF-1α expression in GSC at clinically
achievable concentrations (10–25 nM), it required higher concentration than those
used for cardiac therapy (2–3 nM);
Increased cell
death at a concentration of 10 nM, while 1–5 nM did not reproduce a
significant effect.
|
-
|
[48,49]
|
Atorvastatin
|
10− 4
M significantly decreased cell viability of U 87 and microglia; 10 μM reduced the
invasion and migration of U 87 spheroid cells after 58h.
|
-
|
[50–52]
|
Lovastatin
|
There were 26,
51, 58 and 71% cell death induced by 1, 5, 20, and 40 μM lovastatin alone in M059K
GBM cells, respectively; A significant increase in cell population at G0/G1
phase was observed when cells were treated with 20 μM lovastatin, indicating
that lovastatin was able to arrest the cells at G0/G1 stage.
|
-
|
[50,53,54]
|
Simvastatin
|
10 μM was a
cytotoxic concentration of simvastatin; 10 μM significantly reduced the
number of U 251 and U 87) colonies per
field (pro-apoptotic effect); the survival rates (C6 glioma cells) on
exposure to 2.5, 5, 10, and 20 μM of simvastatin were 96.17, 53.82, 1.76 and 0.49%,
respectively, at 72 h; the survival rates of U 251 cells on exposure to 2.5,
5, 10, and 20 μM of simvastatin were 65.57, 57.59, 25.11 and
21.87%, respectively, at 72 h;
|
-
|
[50,53,55]
|
Mevastatin, fluvastatin
|
5 μM was the cytotoxic concentration of mevastatin and fluvastatin; the
survival rates of C6 cells on exposure to 1, 2.5, 5, and 10 μM of fluvastatin were 69.70, 54.71, 9.71 and
0.88%, respectively, at 72 h; the survival rates of C6 cells on exposure to
1, 2.5, 5, and 10 μM of mevastatin were 83.82, 58.23, 4.41, and 0.52,
respectively, at 72 h; the survival rates of U 251 cells on exposure to 1,
2.5, 5, and 10 μM of mevastatin were 81.44,
58.41, 31.81 and 16.93%, respectively, at 72 h; the survival rates of U 251
cells on exposure to 1, 2.5, 5, and 10 μM of fluvastatin were 63.37, 53.71, 25.45 and 24.08%, respectively, at 72
h.
|
0.922 μM (A 172)
|
[50,53]
|
Cerivastatin
|
50 nM induced mild
morphological changes in U251 cells; 10 μM stimulated the G1 phase arrest; low concentrations (10 and 50 nM)
dose-dependently inhibited the formation of focal adhesion plaques;
disorganized phalloidin staining of actin stress fibers was also noted; at
concentrations from 10 to 100 nM, cerivastatin drastically reduced FAK
phosphorylation at Py397; 1 mg kg−1 cerivastatin per
day intraperitoneally significantly delayed subcutaneous U 87 tumor
growth (average tumour size decreased by 50.2%).
|
0.098 μM (A 172)
|
[50,56]
|
Pitavastatin
|
The IC50 was less
than 10 μM in most of U 87 human GBM
cells tested (range of 1.260 to 55.63 μM); The ability of pitavastatin to cross the BBB is predicted to be
limited as the –log BB was calculated as ‑0.6499; 1 mg kg−1
pitavastatin per day
intraperitoneally significantly delayed subcutaneous U 87 tumor growth (tumor size
decreased by 74.3%).
|
0.334 μM (A172 cells)
21.2, 7.30 and 4.80 μM (U87, 2-, 3- and 4-day
treatments, respectively)
|
[50,57,58]
|
Mibefradil
|
2.5–5 μmol/L
significantly inhibited cell growth and enhanced the inhibition of GSC growth
by TMZ; mibefradil (24 mg/kg bodyweight) was administered per oral gavage (GSC-based xenograft mouse model) every 6 hours for 4
days and resulted in significant inhibition of tumor growth.
|
-
|
[59,60]
|
Captopril
|
MMP-2 and MMP-9 activity was
reduced to half at captopril concentrations of 30–50 nM, levels easily
clinically achieved in humans.
|
-
|
[61]
|
Metformin
|
10 mM
significantly decreased GBM cell proliferation (U 87, U 251, LN18 and SF767);
metformin EC50, as CLIC1 inhibitor, was 2.1 mM, while IAA94
(a well-characterized CLIC1 inhibitor) showed EC50 32 μM (in U 87 cells);
metformin time-dependently decreased U 87 cell viability (EC50:
23, 6.6 and 1.7 mM after 24, 48 and 72 hours); metformin dose-dependently
reduced CSC viability (EC50: 3.9, 11.3, and 8.0 mM for GBM CSC,
after 48 hours of treatment); metformin inhibited CLIC1 conductance in wt GBM
CSCs with an EC50 (2.3 mM) similar to U 87 cells; metformin
(200-1000 μM) significantly inhibited CSC CLIC1 current during
high frequency stimulation (7 days); prolonged treatment (up to 15 days) with
low doses of metformin (10‑300 μM) significantly reduced CSC viability.
|
-
|
[62,63]
|
Repaglinide
|
10 μM inhibited LN229 cell migration (at a much lower concentration compared
to its IC50). Repaglinide (1.04 mg/kg) was administrated daily via
intraperitoneal injection after GBM cell implantation, resulting in a
significant increase in the median survival time (38 days) of mice.
|
200 μM (LN229)
|
[64]
|
Pioglitazone
|
100-200 μM significantly reduced the cellular viability
of glioma cells (U 251, T98G, and U 87) in a concentration- and
time-dependent manner; 100 μM pioglitazone inhibited U 251 cell migration by reduction of MMP-2
expression; 50 μM reduced significantly the metabolic activity of G144 cell
lines; 10 μM promoted only a slight decrease of the metabolic activity in GliNS2
cell line. IC90 value reported for pioglitazone was reported to be
158 μM in U 87 cells.
|
85 μM (U 87)
|
[65,66]
|
Rosiglitazone
|
5–20 μM decreased survival of glioma cells (C6 and U 251) without affecting
primary astrocytes; 50 μM had a small effect on the growth of A 172 and U 87
cells; 50 μM produced only a slight and reversible block in the G2/M phase (M059K cells) at 24 h, which was lost by 48 h.
|
20-30 μM (M059K and M059J)
|
[67,68]
|
Ciglitazone
|
20 μM was toxic
for glioma cells and primary astrocytes; 10 μM was cytoprotective for
primary astrocytes but toxic to glioma cells.
|
-
|
[68]
|
Phenformin
|
100 μM
significantly decreased the proliferation of HF2414 GSCs and their self renewal
(such effect was also observed at a concentration of 50 μM); phenformin (100 μM) inhibited the
expression of the stemness markers OCT4, SOX2 and CD44; phenformin (1 mg/ml)
administered orally in mice harboring GSC-derived xenografts for 4 weeks significantly decreased tumor growth (similar results were
obtained when phenformin (50 mg/kg/day) was administered by intraperitoneal
injection).
|
-
|
[69]
|
Sulfasalazine
|
Concentrations
over 0.5 mM reduced NF-κB activity for prolonged incubation (48 and 72 h);
even at low doses (0.25 mM), sulfasalazine was able to suppress glioma growth
by over 60%;
|
-
|
[70]
|
Aprepitant
|
Maximum inhibition was
reached with aprepitant at 70 μM when no living cells were observed after 48 h of co-culture (GAMG
glioma cell line); 15 μM exerted a growth inhibition 6.71 % in that cell line; the IC50
of aprepitant for non-tumor cells is 90 µM, more than two-fold higher than
for tumor cells.
|
32 µM (GAMG)
|
[71]
|
Cimetidine
|
Significantly
decreased growth rates of both U 373 GBM and 9L gliosarcoma cells at concentrations
equal or higher than 100 mM; 100 and 1000 nM cimetidine significantly
decreased the migration levels of both cell lines; doses between 100 and 0.1
mM induced no modification in either cell cycle kinetics or apoptotic
features.
|
-
|
[72,73]
|
Estradiol
|
In the range of 0.1–25 μM, estradiol decreased cell viability in a concentration-dependent
manner.
|
3.5 μM (C6)
3.8 μM (T98G)
|
[74]
|
Celecoxib
|
In U 87 cells,
celecoxib (8 and 30 μM) significantly induced DNA damage and inhibited
DNA synthesis, corresponding with p53 activation.
|
-
|
[75]
|
Amlexanox
|
150 μM caused a significant increase in G0/G1 and decrease in the S and G2/M
populations of glioma cells, reducing as well the migration capability of
cells; amlexanox intraperitoneally injected, every day for 21 days, in subcutaneous glioma model using U 87 cells, resulted in inhibitory effect
on tumor growth and significantly decreased the tumor volume.
|
120 μM (U 87)
140 μM (U 251)
|
[76]
|
Ivermectin
|
1 μM, 5 μM, and 10 μM inhibited
proliferation of U 87 and T98G cells in a dose-dependent manner:
1 μM, 5 μM, and 10 μM (with ED50
of ∼5 μM); ivermectin at 10 μM completely abolished the
ability of HBMEC to form tubular structures; the serum concentration of the
ivermectin 15 mg/kg administered orally was ∼33 μM; ivermectin
(40 mg/kg) given intraperitoneally during 3 weeks in SCID mouse resulted in
notorious inhibition of U 87 and T98G tumors growth. EC50 value
for ivermectin was reported to be 5 μM.
|
-
|
[77]
|
MTT: 3-(4, 5-dimethylthiazol-2-yl)-2,
5-diphenyltetrazolium bromide; GBM: glioblastoma multiforme; TMZ: temozolomide;
TMZR: temozolomide resistant; MMF: mono-methyl fumarate; TRAIL:
tumor necrosis factor-related apoptosis-inducing ligand; GSC: glioma stem cells;
HIF: hypoxia-inducible factor; FAK: focal adhesion kinase; IC50:
half maximal inhibitory concentration; BBB: blood-brain barrier; MMP: matrix
metalloproteinase; CLIC: chloride intracellular channel; CSC: cancer stem cell;
NF-κB: factor nuclear kappa B; ED50: median
effective dose; HBMEC: human brain microvascular endothelial cells; DNA:
deoxyribonucleic acid; SCID: severe combined immunodeficiency.
References
[1] L.-O.
Roy, M.-B. Poirier, D. Fortin, Chloroquine inhibits the malignant phenotype of
glioblastoma partially by suppressing TGF-beta, Investigational New Drugs. 33
(2015) 1020–1031.
[2] E.B.
Golden, H.-Y. Cho, F.M. Hofman, S.G. Louie, A.H. Schonthal, T.C. Chen,
Quinoline-based antimalarial drugs: a novel class of autophagy inhibitors,
Neurosurgical Focus. 38 (2015) E12.
[3] P. Erkoc,
A. Cingöz, T.B. Onder, S. Kizilel, Quinacrine Mediated Sensitization of
Glioblastoma (GBM) Cells to TRAIL through MMP-Sensitive PEG Hydrogel Carriers,
Macromolecular Bioscience. 17 (2017) 1-13.
[4] C.
Venugopal, R. Hallett, P. Vora, B. Manoranjan, S. Mahendram, M.A. Qazi, N.
McFarlane, M. Subapanditha, S.M. Nolte, M. Singh, D. Bakhshinyan, N. Garg, T.
Vijayakumar, B. Lach, J.P. Provias, K. Reddy, N.K. Murty, B.W. Doble, M.
Bhatia, J.A. Hassell, S.K. Singh, Pyrvinium Targets CD133 in Human Glioblastoma
Brain Tumor-Initiating Cells, Clinical Cancer Research. 21 (2015) 5324-5337.
[5] R.Y. Bai,
V. Staedtke, C.M. Aprhys, G.L. Gallia, G.J. Riggins, Antiparasitic mebendazole
shows survival benefit in 2 preclinical models of glioblastoma multiforme,
Neuro-Oncology. 13 (2011) 974–982.
[6] S.L.
Kominsky, P.S. Subramaniam, H.M. Johnson, B.A. Torres, Inhibitory effects of
IFN-gamma and acyclovir on the glioblastoma cell cycle., Journal of Interferon
& Cytokine Research. 20 (2000) 463–469.
[7] M.S.
Ahluwalia, C. Patton, G. Stevens, T. Tekautz, L. Angelov, M.A. Vogelbaum, R.J.
Weil, S. Chao, P. Elson, J.H. Suh, G.H. Barnett, D.M. Peereboom, Phase II trial
of ritonavir/lopinavir in patients with progressive or recurrent high-grade
gliomas., Journal of Neuro-Oncology. 102 (2011) 317–321.
[8] N.
Laurent, S. de Bouard, J.-S. Guillamo, C. Christov, R. Zini, H. Jouault, P.
Andre, V. Lotteau, M. Peschanski, Effects of the proteasome inhibitor ritonavir
on glioma growth in vitro and in vivo, Molecular Cancer Therapeutics. 3 (2004)
129–136.
[9] P. Pyrko,
A. Kardosh, W. Wang, W. Xiong, A.H. Schonthal, T.C. Chen, HIV-1 protease
inhibitors nelfinavir and atazanavir induce malignant glioma death by
triggering endoplasmic reticulum stress, Cancer Research. 67 (2007)
10920–10928.
[10] A. Ogino,
E. Sano, Y. Ochiai, S. Yamamuro, S. Tashiro, K. Yachi, T. Ohta, T. Fukushima,
Y. Okamoto, K. Tsumoto, T. Ueda, A. Yoshino, Y. Katayama, Efficacy of ribavirin
against malignant glioma cell lines, Oncology Letters. 8 (2014) 2469–2474.
[11] R. Liu, J.
Li, T. Zhang, L. Zou, Y. Chen, K. Wang, Y. Lei, K. Yuan, Y. Li, J. Lan, L.
Cheng, N. Xie, R. Xiang, E.C. Nice, C. Huang, Y. Wei, Itraconazole suppresses
the growth of glioblastoma through induction of autophagy: involvement of
abnormal cholesterol trafficking, Autophagy. 10 (2014) 1241–1255.
[12] A. Zandi,
T.M. Zanjani, S.A. Ziai, Y.K. Poul, M. Haji, M. Hoseini, Evaluation of the
Cytotoxic Effects of Ciprofloxacin on Human Glioblastoma, 8 (2017) 119–126.
[13] F. Atif,
N.R. Patel, S. Yousuf, D.G. Stein, The Synergistic Effect of Combination
Progesterone and Temozolomide on Human Glioblastoma Cells, PLOS One. 10 (2015)
e0131441.
[14] C.
Naujokat, R. Steinhart, Salinomycin as a drug for targeting human cancer stem
cells, Journal of Biomedicine and Biotechnology. 2012 (2012) 1-17.
[15] W.-T. Liu,
C.-H. Lin, M. Hsiao, P.-W. Gean, Minocycline inhibits the growth of glioma by
inducing autophagy, Autophagy. 7 (2011) 166–175.
[16] A. Wang-Gillam,
E. Siegel, D. a Mayes, L.F. Hutchins, Anti-Tumor Effect of Doxycycline on
Glioblastoma Cells, Journal of Cancer Molecules. 3(5) (2007) 147–153.
[17] R. Yang,
L. Yi, Z. Dong, Q. Ouyang, J. Zhou, Y. Pang, Y. Wu, L. Xu, H. Cui, Tigecycline
Inhibits Glioma Growth by Regulating miRNA-199b-5p-HES1-AKT Pathway, Molecular
Cancer Therapeutics. 15 (2016) 421–429.
[18] C.R.
Oliva, W. Zhang, C. Langford, M.J. Suto, C.E. Griguer, Repositioning
chlorpromazine for treating chemoresistant glioma through the inhibition of
cytochrome c oxidase bearing the COX4-1 regulatory subunit, Oncotarget. 8
(2017) 37568–37583.
[19] J.K. Lee,
D.H. Nam, J. Lee, Repurposing antipsychotics as glioblastoma therapeutics:
Potentials and challenges (Review), Oncology Letters. 11 (2016) 1281–1286.
[20] H.-W.
Cheng, Y.-H. Liang, Y.-L. Kuo, C.-P. Chuu, C.-Y. Lin, M.-H. Lee, A.T.H. Wu,
C.-T. Yeh, E.I.-T. Chen, J. Whang-Peng, C.-L. Su, C.-Y.F. Huang, Identification
of thioridazine, an antipsychotic drug, as an antiglioblastoma and anticancer
stem cell agent using public gene expression data, Cell Death & Disease. 6
(2015) e1753.
[21] I. Gil-Ad,
B. Shtaif, Y. Levkovitz, M. Dayag, E. Zeldich, A. Weizman, Characterization of
phenothiazine-incluced apoptosis in neuroblastoma and glioma cell lines:
Clinical relevance and possible application for brain-derived tumors, Journal
of Molecular Neuroscience. 22 (2004) 189–198.
[22] R.E. Kast,
B.M. Ellingson, C. Marosi, M.-E. Halatsch, Glioblastoma treatment using
perphenazine to block the subventricular zone’s tumor trophic functions,
Journal of Neuro-Oncology. 116 (2014) 207–212.
[23] S. Tzadok,
E. Beery, M. Israeli, O. Uziel, M. Lahav, E. Fenig, I. Gil-Ad, A. Weizman, J.
Nordenberg, In vitro novel combinations of psychotropics and anti-cancer
modalities in U87 human glioblastoma cells, International Journal of Oncology.
37 (2010) 1043–1051.
[24] G.
Karpel-Massler, R.E. Kast, M.A. Westhoff, A. Dwucet, N. Welscher, L.
Nonnenmacher, M. Hlavac, M.D. Siegelin, C.R. Wirtz, K.M. Debatin, M.E.
Halatsch, Olanzapine inhibits proliferation, migration and
anchorage-independent growth in human glioblastoma cell lines and enhances
temozolomide’s antiproliferative effect, J Neuro-Oncology. 122 (2015) 21–33.
[25] A. Ranjan,
S.K. Srivastava, Penfluridol suppresses glioblastoma tumor growth by
Akt-mediated inhibition of GLI1, Oncotarget. 8 (2017) 32960–32976.
[26] Y. Wang,
N. Huang, H. Li, S. Liu, X. Chen, S. Yu, N. Wu, X.-W. Bian, H.-Y. Shen, C. Li,
L. Xiao, Promoting oligodendroglial-oriented differentiation of glioma stem
cell: a repurposing of quetiapine for the treatment of malignant glioma,
Oncotarget. 8 (2017) 37511–37524.
[27] M.O.
Nowicki, N. Dmitrieva, A.M. Stein, J.L. Cutter, J. Godlewski, Y. Saeki, M.
Nita, M.E. Berens, L.M. Sander, H.B. Newton, E.A. Chiocca, S. Lawler, Lithium
inhibits invasion of glioma cells; possible involvement of glycogen synthase
kinase-3, Neuro-Oncology. 10 (2008) 690–699.
[28] S. Han, L.
Meng, Y. Jiang, W. Cheng, X. Tie, J. Xia, A. Wu, Lithium enhances the
antitumour effect of temozolomide against TP53 wild-type glioblastoma cells via
NFAT1/FasL signalling, British Journal of Cancer. 116 (2017) 1302–1311.
[29] E.G. Shaw,
R. Rosdhal, R.B.J. D’Agostino, J. Lovato, M.J. Naughton, M.E. Robbins, S.R.
Rapp, Phase II study of donepezil in irradiated brain tumor patients: effect on
cognitive function, mood, and quality of life, Journal of Clinical Oncology. 24
(2006) 1415-1420.
[30] V.
Megalizzi, C. Decaestecker, O. Debeir, S. Spiegl-Kreinecker, W. Berger, F.
Lefranc, R.E. Kast, R. Kiss, Screening of anti-glioma effects induced by
sigma-1 receptor ligands: potential new use for old anti-psychiatric medicines,
European Journal of Cancer (Oxford, England : 1990). 45 (2009) 2893–2905.
[31] W.-S.
Yoon, M.-Y. Yeom, E.-S. Kang, Y.-A. Chung, D.-S. Chung, S.-S. Jeun, Memantine
Induces NMDAR1-Mediated Autophagic Cell Death in Malignant Glioma Cells,
Journal of Korean Neurosurgical Society. 60 (2017) 130–137.
[32] Y.
Levkovitz, I. Gil-Ad, E. Zeldich, M. Dayag, A. Weizman, Differential induction
of apoptosis by antidepressants in glioma and neuroblastoma cell lines:
evidence for p-c-Jun, cytochrome c, and caspase-3 involvement, Journal of
Molecular Neuroscience. 27 (2005) 29–42.
[33] K. Liu, S.
Yang, Y.-K. Lin, J.-W. Lin, Y. Lee, J.-Y. Wang, C. Hu, E. Lin, S. Chen, C.
Then, S. Shen, Fluoxetine, an antidepressant, suppresses glioblastoma by
evoking AMPAR-mediated calcium-dependent apoptosis, Oncotarget. 6 (2015)
5088–101.
[34] K.
Hayashi, H. Michiue, H. Yamada, K. Takata, H. Nakayama, F.-Y. Wei, A. Fujimura,
H. Tazawa, A. Asai, N. Ogo, H. Miyachi, T. Nishiki, K. Tomizawa, K. Takei, H.
Matsui, Fluvoxamine, an anti-depressant, inhibits human glioblastoma invasion
by disrupting actin polymerization, Scientific Reports. 6 (2016) 23372.
[35] S.-H.
Jeon, S.H. Kim, Y. Kim, Y.S. Kim, Y. Lim, Y.H. Lee, S.Y. Shin, The tricyclic
antidepressant imipramine induces autophagic cell death in U-87MG glioma cells,
Biochemical and Biophysical Research Communications. 413 (2011) 311–317.
[36] K.
Shchors, A. Massaras, D. Hanahan, Dual Targeting of the Autophagic Regulatory
Circuitry in Gliomas with Repurposed Drugs Elicits Cell-Lethal Autophagy and
Therapeutic Benefit, Cancer Cell. 28 (2015) 456–471.
[37] A.M.
Bielecka-Wajdman, M. Lesiak, T. Ludyga, A. Sieroń, E. Obuchowicz, Reversing
glioma malignancy: a new look at the role of antidepressant drugs as adjuvant
therapy for glioblastoma multiforme, Cancer Chemotherapy and Pharmacology. 79
(2017) 1249–1256.
[38] A.M. Bielecka-Wajdman,
M. Lesiak, T. Ludyga, A. Sieron, E. Obuchowicz, Reversing glioma malignancy: a
new look at the role of antidepressant drugs as adjuvant therapy for
glioblastoma multiforme., Cancer Chemotherapy and Pharmacology. 79 (2017)
1249–1256.
[39] D.
Rundle-Thiele, R. Head, L. Cosgrove, J.H. Martin, Repurposing some older drugs
that cross the blood-brain barrier and have potential anticancer activity to
provide new treatment options for glioblastoma, British Journal of Clinical
Pharmacology. 81 (2016) 199–209.
[40] M. Dikmen,
Z. Canturk, Y. Ozturk, Escitalopram oxalate, a selective serotonin reuptake
inhibitor, exhibits cytotoxic and apoptotic effects in glioma C6 cells, Acta
Neuropsychiatrica. 23 (2011) 173–178.
[41] A.
Marutani, M. Nakamura, F. Nishimura, T. Nakazawa, R. Matsuda, Y. Hironaka, I.
Nakagawa, K. Tamura, Y. Takeshima, Y. Motoyama, E. Boku, Y. Ouji, M. Yoshikawa,
H. Nakase, Tumor-inhibition effect of levetiracetam in combination with
temozolomide in glioblastoma cells, Neurochemical Journal. 11 (2017) 43–49.
[42] J.-H.
Tseng, C.-Y. Chen, P.-C. Chen, S.-H. Hsiao, C.-C. Fan, Y.-C. Liang, C.-P. Chen,
Valproic acid inhibits glioblastoma multiforme cell growth via paraoxonase 2
expression, Oncotarget. 8 (2017) 14666–14679.
[43] M. Eckert,
L. Klumpp, S. Huber, Cellular Effects of the Antiepileptic Drug Valproic Acid
in Glioblastoma, Cellular Physiology and Biochemistry. 44 (2017) 1591–1605.
[44] S.S. Hsu,
C.R. Jan, W.Z. Liang, Evaluation of cytotoxicity of propofol and its related
mechanism in glioblastoma cells and astrocytes, Environmental Toxicology. 32
(2017) 2440–2454.
[45] P. Liu, S.
Brown, T. Goktug, P. Channathodiyil, V. Kannappan, J.P. Hugnot, P.O. Guichet,
X. Bian, A.L. Armesilla, J.L. Darling, W. Wang, Cytotoxic effect of
disulfiram/copper on human glioblastoma cell lines and ALDH-positive
cancer-stem-like cells, British Journal of Cancer. 107 (2012) 1488–1497.
[46] J.
Triscott, C. Lee, K. Hu, A. Fotovati, R. Berns, M. Pambid, M. Luk, R.E. Kast,
E. Kong, E. Toyota, S. Yip, B. Toyota, S.E. Dunn, Disulfiram, a drug widely
used to control alcoholism, suppresses the self-renewal of glioblastoma and
over-rides resistance to temozolomide, Oncotarget. 3 (2012) 1112–1123.
[47] L. Booth,
N. Cruickshanks, S. Tavallai, J.L. Roberts, M. Peery, A. Poklepovic, P. Dent,
Regulation of dimethyl-fumarate toxicity by proteasome inhibitors, Cancer
Biology & Therapy. 15 (2014) 1646–1657.
[48] D.-H. Lee,
S.C. Oh, A.J. Giles, J. Jung, M.R. Gilbert, D.M. Park, Cardiac glycosides
suppress the maintenance of stemness and malignancy via inhibiting HIF-1α in human glioma stem cells, Oncotarget. 8 (2017)
40233–40245.
[49] Z. Shang,
L. Zhang, Digitoxin increases sensitivity of glioma stem cells to
TRAIL-mediated apoptosis, Neuroscience Letters. 653 (2017) 19–24.
[50] P. Jiang,
R. Mukthavaram, Y. Chao, N. Nomura, I.S. Bharati, V. Fogal, S. Pastorino, D.
Teng, X. Cong, S.C. Pingle, S. Kapoor, K. Shetty, A. Aggrawal, S. Vali, T.
Abbasi, S. Chien, S. Kesari, In vitro and in vivo anticancer effects of
mevalonate pathway modulation on human cancer cells, British Journal of Cancer.
111 (2014) 1562–1571.
[51] Y.
Yongjun, H. Shuyun, C. Lei, C. Xiangrong, Y. Zhilin, K. Yiquan, Atorvastatin
suppresses glioma invasion and migration by reducing microglial MT1-MMP
expression, Journal of Neuroimmunology. 260 (2013) 1–8.
[52] N. Bayat,
S. Ebrahimi-Barough, A. Norouzi-Javidan, H. Saberi, R. Tajerian, M.M.M.
Ardakan, S. Shirian, A. Ai, J. Ai, Apoptotic effect of atorvastatin in
glioblastoma spheroids tumor cultured in fibrin gel, Biomedicine &
Pharmacotherapy. 84 (2016) 1959–1966.
[53] M. Yanae,
M. Tsubaki, T. Satou, T. Itoh, M. Imano, Y. Yamazoe, S. Nishida, Statin-induced
apoptosis via the suppression of ERK1/2 and Akt activation by inhibition of the
geranylgeranyl-pyrophosphate biosynthesis in glioblastoma., Journal of
Experimental & Clinical Cancer Research. 30 (2011) 1-8.
[54] D.Y.L. Chan, G.G. Chen, W.S. Poon, P.C. Liu,
Lovastatin sensitized human glioblastoma cells to TRAIL-induced apoptosis,
Journal of Neuro-Oncology. 86 (2008) 273–283.
[55] H. Wu, H.
Jiang, D. Lu, Y. Xiong, C. Qu, D. Zhou, A. Mahmood, M. Chopp, Effect of
simvastatin on glioma cell proliferation, migration, and apoptosis,
Neurosurgery. 65 (2009) 1087-1097.
[56] S. Obara,
M. Nakata, H. Takeshima, J. Kuratsu, I. Maruyama, I. Kitajima, Inhibition of
migration of human glioblastoma cells by cerivastatin in association with focal
adhesion kinase (FAK), Cancer Letters. 185 (2002) 153–161.
[57] M.I.
Koukourakis, A.G. Mitrakas, A. Giatromanolaki, Therapeutic interactions of
autophagy with radiation and temozolomide in glioblastoma: evidence and issues
to resolve, British Journal of Cancer. 114 (2016) 485–496.
[58] P. Jiang,
R. Mukthavavam, Y. Chao, I.S. Bharati, V. Fogal, S. Pastorino, X. Cong, N.
Nomura, M. Gallagher, T. Abbasi, S. Vali, S.C. Pingle, M. Makale, S. Kesari,
Novel anti-glioblastoma agents and therapeutic combinations identified from a
collection of FDA approved drugs, Journal of Translational Medicine. 12 (2014)
1–13.
[59] Y. Zhang,
N. Cruickshanks, F. Yuan, B. Wang, M. Pahuski, J. Wulfkuhle, I. Gallagher, A.F.
Koeppel, S. Hatef, C. Papanicolas, J. Lee, E.E. Bar, D. Schiff, S.D. Turner,
E.F. Petricoin, L.S. Gray, R. Abounader, Targetable T-type calcium channels
drive glioblastoma, Cancer Research. 77 (2017) 3479–3490.
[60] S.T. Keir,
H.S. Friedman, D.A. Reardon, D.D. Bigner, L.A. Gray, Mibefradil, a novel
therapy for glioblastoma multiforme: cell cycle synchronization and interlaced
therapy in a murine model, Journal of Neuro-Oncology. 111 (2013) 97–102.
[61] R.E. Kast,
M.E. Halatsch, Matrix Metalloproteinase-2 and -9 in Glioblastoma: A Trio of Old
Drugs-Captopril, Disulfiram and Nelfinavir-Are Inhibitors with Potential as
Adjunctive Treatments in Glioblastoma, Archives of Medical Research. 43 (2012)
243–247.
[62] J. Sesen,
P. Dahan, S.J. Scotland, E. Saland, V.T. Dang, A. Lemarié, B.M. Tyler, H. Brem,
C. Toulas, E.C.J. Moyal, J.E. Sarry, N. Skuli, Metformin inhibits growth of
human glioblastoma cells and enhances therapeutic response, PLOS ONE. 10 (2015)
1–24.
[63] S. Ho
Yang, S. Li, G. Lu, H. Xue, D.H. Kim, J.-J. Zhu, Y. Liu, S. Ho Yang, S. Li, G.
Lu, H. Xue, D.H. Kim, J.-J. Zhu, Y. Liu, Metformin treatment reduces
temozolomide resistance of glioblastoma cells, Oncotarget. 7 (2016)
78787–78803.
[64] Z.X. Xiao,
R.Q. Chen, D.X. Hu, X.Q. Xie, S. Bin Yu, X.Q. Chen, Identification of
repaglinide as a therapeutic drug for glioblastoma multiforme, Biochemical and
Biophysical Research Communications. 488 (2017) 33–39.
[65] Z. Wan, W.
Shi, B. Shao, J. Shi, A. Shen, Y. Ma, J. Chen, Q. Lan, Peroxisome
proliferator-activated receptor gamma agonist pioglitazone inhibits
beta-catenin-mediated glioma cell growth and invasion, Molecular and Cellular
Biochemistry. 349 (2011) 1–10.
[66] C.
Cilibrasi, V. Butta, G. Riva, A. Bentivegna, Pioglitazone Effect on Glioma Stem
Cell Lines: Really a Promising Drug Therapy for Glioblastoma?, PPAR Research.
2016 1-9.
[67] R.
Morosetti, T. Servidei, M. Mirabella, S. Rutella, A. Mangiola, G. Maira, R.
Mastrangelo, H.P. Koeffler, The PPARgamma ligands PGJ2 and rosiglitazone show a
differential ability to inhibit proliferation and to induce apoptosis and
differentiation of human glioblastoma cell lines, International Journal of
Oncology. 25 (2004) 493–502.
[68] J.M. Perez-Ortiz, P. Tranque,
C.F. Vaquero, B. Domingo, F. Molina, S. Calvo, J. Jordan, V. Cena, J. Llopis,
Glitazones differentially regulate primary astrocyte and glioma cell survival. Involvement of reactive oxygen species and peroxisome
proliferator-activated receptor-gamma, The Journal of Biological Chemistry. 279
(2004) 8976–8985.
[69] W. Jiang,
S. Finniss, S. Cazacu, C. Xiang, Z. Brodie, L. Poisson, D.B. Shackelford, C.
Brodie, Repurposing phenformin for the targeting of glioma stem cells and the
treatment of glioblastoma, Oncotarget. 7 (2016) 56456-56470.
[70] L. Sleire,
B.S. Skeie, I.A. Netland, H.E. Førde, E. Dodoo, F. Selheim, L. Leiss, J.I.
Heggdal, P.H. Pedersen, J. Wang, P. Enger, Drug repurposing: Sulfasalazine
sensitizes gliomas to gamma knife radiosurgery by blocking cystine uptake
through system Xc-, leading to glutathione depletion, Oncogene. 34 (2015)
5951–5959.
[71] R.E. Kast,
S. Ramiro, S. Llado, S. Toro, R. Covenas, M. Munoz, Antitumor action of
temozolomide, ritonavir and aprepitant against human glioma cells, Journal of
Neuro-Oncology. 126 (2016) 425–431.
[72] F. Lefranc,
S. James, I. Camby, J.-F. Gaussin, F. Darro, J. Brotchi, J. Gabius, R. Kiss,
Combined cimetidine and temozolomide, compared with temozolomide alone:
significant increases in survival in nude mice bearing U373 human glioblastoma
multiforme orthotopic xenografts, Journal of Neurosurgery. 102 (2005) 706–714.
[73] T. Furuta,
H. Sabit, D. Yu, K. Miyashita, M. Kinoshita, N. Uchiyama, Y. Hayashi, Y.
Hayashi, T. Minamoto, M. Nakada, T. Furuta, H. Sabit, Y. Dong, K. Miyashita, M.
Kinoshita, N. Uchiyama, Y. Hayashi, Y. Hayashi, T. Minamoto, M. Nakada, T.
Furuta, H. Sabit, D. Yu, K. Miyashita, M. Kinoshita, N. Uchiyama, Y. Hayashi,
Y. Hayashi, T. Minamoto, M. Nakada, Biological basis and clinical study of
glycogen synthase kinase- 3β-targeted
therapy by drug repositioning for glioblastoma, Oncotarget. 8 (2017)
22811–22824.
[74] N. Altiok,
M. Ersoz, M. Koyuturk, Estradiol induces JNK-dependent apoptosis in
glioblastoma cells, Oncology Letters. 2 (2011) 1281–1285.
[75] K.B. Kang,
C. Zhu, S.K. Yong, Q. Gao, M.C. Wong, Enhanced sensitivity of celecoxib in
human glioblastoma cells: Induction of DNA damage leading to p53-dependent G(1)
cell cycle arrest and autophagy, Molecular Cancer. 8 (2009) 1-16.
[76] Y. Liu, J.
Lu, Z. Zhang, L. Zhu, S. Dong, G. Guo, R. Li, Y. Nan, K. Yu, Y. Zhong, Q.
Huang, Amlexanox, a selective inhibitor of IKBKE, generates anti-tumoral
effects by disrupting the Hippo pathway in human glioblastoma cell lines, Cell
Death & Disease. 8 (2017) e3022.
[77] Y. Liu, S.
Fang, Q. Sun, B. Liu, Anthelmintic drug ivermectin inhibits angiogenesis,
growth and survival of glioblastoma through inducing mitochondrial dysfunction
and oxidative stress, Biochemical and Biophysical Research Communications. 480
(2016) 415–421.